Friday, March 19, 2010
The Nephrologist as Team Leader
In order to prepare for the eventuality of one of these surveys, I received a memo this week with a reminder of my responsibilities as the Hemodialysis Unit Medical Director:
"By virtue of the Medical Director's leadership of the facility's Governing Body, his/her responsibilities extend beyond the primary focus on improving quality of care delivered in the facility and extends to the general management and operations of the facility. As such, all condition level citations are linked to /and are the responsibility of the Medical Director according to CMS".
It went on to append a summary of findings from recent audits, occurring in a particular geographical region, identifying some of the deficiencies which have been noted by the auditors as citable or worthy of a conditional warning. Here are a few of them:
Infection control standards not met due to:
- Patients not washing and sanitizing their hands pre and post treatment
- Staff members accessing computer terminals with gloves on after patient contact
- Patients self holding needle access sites post treatment without gloves on
Physical environment standards not met due to:
- Unsanitary and non working sinks and faucets
- Open boxes of needles and syringes not properly stored
Documentation requirements not met due to:
- Incomplete Interdisciplinary Team assessments
- Medical records not stored properly
- Peritoneal dialysis orders incompletely and improperly written.
What does this all mean? At first glance these citations/conditions may seem to be out of the realm of the practicing clinician whose focus is elsewhere, but in actuality they are integral to the proper care of our patients and the infection control lapses certainly should raise a high level of concern. We all have a natural disinclination to being surveyed but maybe these surveys are useful if they encourage us to put into place processes which ensure these lapses do not occur and if we develop systems of self assessment and quality improvement within our domain of responsibility.
Which brings me to the point of this blog. It's all about the team, and as Physicians we assume a natural leadership role when we deliver care to our patients and some of us are eventually appointed to leadership positions as our career advances. The usual dialysis unit personnel resources include Nurses, Patient Care Technologists, Dieticians, Social Workers and Administrative Staff. CMS is telling us we are responsible for the performance of this team on multiple levels. Traditionally our training has not focused much attention on preparing us for such leadership. But, the ability to develop and lead a team in the delivery of care to a defined patient population such as an office practice or a dialysis facility may be as important as an extensive clinical knowledge base in terms of improving outcomes. Being able to function as a clinician and a team leader is a challenge and interestingly many of the leadership skills we eventually acquired are done so by osmosis and without direct teaching.
For those interested the Medicare and Medicaid Programs; Conditions for Coverage for End-Stage Renal Disease Facilities; Final Rule can be found in the Federal Register / Vol. 73, No. 73 / Tuesday, April 15, 2008 / Rules and Regulations. It is ~ 120 pages but is worth reviewing if you are planning career involving outpatient dialysis care.
Posted by David Steele M.D.
Thursday, March 18, 2010
HCG in Kidney Disease
 That cardiac biomarkers, including Troponin and BNP, can be elevated in CKD or ESKD without necessarily having clinical significance is familiar to most of us. Whether you believe that an elevated Troponin in ESKD is due to diminished clearance or some constant low-level myocardial damage of unclear significance, most would be comfortable dismissing borderline positive results without suggestive signs or symptoms.
That cardiac biomarkers, including Troponin and BNP, can be elevated in CKD or ESKD without necessarily having clinical significance is familiar to most of us. Whether you believe that an elevated Troponin in ESKD is due to diminished clearance or some constant low-level myocardial damage of unclear significance, most would be comfortable dismissing borderline positive results without suggestive signs or symptoms.How to distinguish? The urine HCG assay does not cross-react with these substances, and so, if the patient still makes urine, confirmatory testing with urine HCG is a reasonable first step. In ESKD patients who no longer make urine, progesterone levels can be used as a next step especially when more immediate knowledge of pregnancy status is required. If progesterone is low, viable pregnancy is unlikely, though serial serum HCG levels should be monitored to rule out non-viable pregnancy (egs, ectopic). If there is no urgency to determining pregnancy status, follow-up serum HCG to see if doubling occurs at expected intervals should be performed. In my patient's case, her serum HCG level one week later was the same as that one week prior, and the serum HCG was deemed to be elevated due to decreased clearance in ESKD.
Wednesday, March 17, 2010
Don't forget to look for proteinuria!

At Renal Grand Rounds yesterday, we discussed the case of a previously healthy 50 year old man who presented with fulminant hepatic failure and AKI. He was initially thought to have AKI related to either SIRS/hypotension or Hepatorenal Syndrome (which should be a diagnosis of exclusion), but was astutely noted to have significant (about 6 gm/day) proteinuria. The patient was worked up further: his serum electrophoresis revealed a monoclonal immunoglobulin spike and urine Bence Jones protein was present. Serum free kappa light chains were high and the kappa to lambda light chain ratio was significantly elevated. Concomitant liver biopsy revealed AL amyloidosis, which was further confirmed and typed by fat pad and colon biopsies. Unfortunately, the patient succumbed to multi-organ failure.
The teaching point from this case is that the proteinuria was the key to making the correct diagnosis.
By way of quick review, amyloidosis is a group of diseases characterized by extracellular deposition of beta sheet fibrils which, in systemic forms, cause progressive organ dysfunction.
AL amyloidosis is characterized by fibrils that contain fragments of the variable portion of monoclonal light chains. This is the result of a clonal expansion of plasma cells, but interestingly, most patients with AL amyloidosis do not develop the manifestations of multiple myeloma, and most multiple myeloma patients do not have amyloid deposition. What both diseases have in common, however, is renal dysfunction which presents with significant proteinuria. Of note, given the pathophysiology of these diseases, the proteinuria is not related to albuminuria. Therefore, it is important to obtain a spot protein to creatinine ratio on all patients who may have otherwise unexplained or difficult to explain AKI, and subsequently pursue further work-up, including free serum light chain measurement.
In AL amyloidosis, response to therapy correlates with reduction in circulating light chains, so a hematologic response is the first marker in anticipation of renal function recovery or improvement. Interestingly, as well, the degree of baseline proteinuria on presentation correlates with response to treatment. Current treatment options include melphalan (an alkylating agent) plus steroids, Velcade (Bortezomib, a proteasome inhibitor) and rescue stem cell transplantation, which remains controversial.
Endocrine and thiazide-induced hyponatremia
The mechanism in hypothyroidism is incompletely understood, but felt to relate mainly to a reduction in cardiac output, increased activity of baroreceptors and subsequent non-osmotic release of ADH. Often, a reduction in GFR is noted in hypothyroid patients, thereby reducing the quantity of free water that reaches the distal nephron. Correction can be achieved by the administration of exogenous thyroid hormone.
In adrenal insufficiency, the reduction in cortisol may cause reduced cardiac output and blood pressure, again via a poorly understood mechanism. This can result in non-osmotic ADH release and tendency towards free water retention. Perhaps more importantly, ADH release from the posterior pituitary is under the control of CRH from the hypothalamus. CRH in turn is under negative feedback control from cortisol. So, in states of low cortisol, the negative feedback on CRH is lost and, in turn, ADH release is stimulated.
Why do thiazides cause more hyponatraemia than loop diuretics?
The efficacy of ADH depends on the generation of the medullary concentration gradient via NaCl reabsorption, without water, in the thick ascending limb of the loop of Henle. This creates a gradient for water reabsorption via aquaporin-2 insertion in the luminal membranes of the cortical and outer medullary collecting tubules.
With this in mind, it becomes clearer why thiazide diuretics are more likely to cause hyponatraemia than loop diuretics. Loop diuretics, by inhibiting the sodium reabsorption in the thick ascending limb of Henle’s loop, interfere with the generation of the hypertonic medullary interstitium; so, despite inducing a mild increase in ADH secondary to volume depletion, the ADH is less effective because there is a reduced concentration gradient for water reabsorption. Thiazide diuretics act on the distal tubule, having no effect on the medullary concentration gradient, thereby allowing ADH induced by volume depletion to promote water reabsorption unabated.
Posted by Finnian McCausland M.D.
Monday, March 15, 2010
VEGF inhibitors and proteinuria
There is a documented association between VEGF inhibitors (e.g. bevacizumab, sunitinib, sorafenib, pazopanib, axitinib) and renal disease. While these agents are usually well tolerated, their toxic effects include proteinuria and hypertension. The incidence of proteinuria has been variable in different studies depending on patient characteristics, cancer type, and signals targeted, though a number of clinical studies report that treatment with bevacizumab (avastin) may be associated with increased proteinuria. For instance, bevacizumab has been associated with proteinuria in 23-38% of patients treated for colorectal cancer and in up to 64% of patients treated for renal cell carcinoma. One meta-analysis of RCTs with patients receiving bevacizumab reports a relative risk of 1.4 for proteinuria at a low dose (2.5 to 7.5 g/kg) and 1.6 for a high dose (10 to 15 mg/kg) of bevacizumab. Proteinuria appears to at least partly resolve with discontinuation of the VEGF inhibitor, though some patients are able to resume therapy without an increase in proteinuria. It is not clear at this time whether there is an association between duration of treatment and the development of proteinuria.
- Collapsing glomerulopathy
- Cryoglobulinemic glomerlonephritis
- Immune complex-associated focal proliferative GN
- Sorafenib-induced acute interstitial nephritis
Of note, this topic of nephrotoxicity associated with VEGF inhibitors has recently made it to the nephrology boards!
Saturday, March 13, 2010
A trap for angiotensin type 1 receptors
 The renin-angiotensin system is powerful regulator of blood pressure homeostasis. An article published in the March 24 issue of JASN highlights an emerging area of research into the modulation of AT1 receptors on different tissues.
The renin-angiotensin system is powerful regulator of blood pressure homeostasis. An article published in the March 24 issue of JASN highlights an emerging area of research into the modulation of AT1 receptors on different tissues.The majority of well-known angiotensin II actions are mediated via AT1 receptor stimulation, and angiotensin converting enzyme inhibitors (ACEi) and AT1 receptor blockers (ARBs) have been widely used as antihypertensive agents with cardiovascular protective effects. Elucidating factors that regulate AT1 receptor expression levels in different tissue compartments will hopefully lead to novel agents to treat hypertension and its associated end-organ damage (ESRD, CHF and Stroke). Recent studies have demonstrated the existence of several proteins interacting with AT1 receptors that may modulate AT1 receptor expression level, sensitivity and internalization. A recent review article highlights this emerging field. Atrap (AT1 receptor-associated protein) is the best characterized protein and the focus of the March 24 article by Oppermann et al.
It was previously reported that Atrap (a 19kD protein) interacts specifically with the carboxyl-terminal domain of the A1a receptor and catalyzes its internalization in cultured cells. Prior in vivo studies over-expressing the Atrap protein in the heart, aortae and femoral artery in mice demonstrated a protective effect in these tissues in response to angiotensin II infusion. Overall, the prevailing literature suggested an inhibitory or protective effect of Atrap on AT1 receptor function. This group knocked out the Atrap gene in mice to test this hypothesis in vivo.
They reported that Atrap KO mice have a higher resting blood pressure (by 10 mmHg) as measured by radiotelemety. Suggesting that Atrap is important in regulating basal blood pressure and that its absence leads to systemic hypertension. Interestingly, they found that Atrap is highly expressed in the kidney (then testis=adrenal>heart>lung=liver=aorta=brain). Specifically, they showed that Atrap is highly expressed in the proximal tubule of the kidney. Previous investigations have focused on the distal tubule as potential sites that affect blood pressure as most of the known genetic mutations in sodium transport causing hyper/hypotension are linked to the distal nephron (Bartter, Little, Gitelman syndromes, etc). This group reports that Atrap KO mice have more AT1 receptors in the proximal tubule which allows for increased angII-dependent NHE-3 activity causing volume expansion and hypertension.
This highlights the importance of investigating novel pathways regulating the renin-angiotensin system. Modulation of this system with the use of ACEi, ARBs and now direct renin inhibitors have continued to be the mainstay of therapy for patients with heart failure, diabetes, hypertension and kidney disease. Activation of Atrap may be more specific and more physiological for inhibition of At1 receptor signaling. New drug discovery modulating Atrap protein expression could lead to novel therapy. For now, much more research is needed.
Friday, March 12, 2010
Mythbusters: Hypertension

It seems like there have been a lot of recent developments in the management of hypertension, and several sacred cows have become burgermeat. I thought I'd summarize a few of these important developments here:
1. Blood pressure targets in the over 80's
The Hypertension in the Very Elderly (HYVET) trial taught us that treating hypertension in the very elderly is effective in preventing stroke. However, we may have been over-treating the "oldest-olds" for years, and that target SBPs for this group should be< 150/80mmHg according to a recent Cochrane review. As nephrologists, we may have been particularly culpable, due inappropriate application of a 130/80 mmHg target for elderly patients with mild reductions in GFR ("CKD").
2. Who benefits from aldosterone blockade?
A strong association has been demonstrated between abdominal obesity, sleep apnea and aldosterone excess, such that it has been suggested that visceral fat may be a source of aldosterone, contributing to resistant hypertension in the abdominally obese. Recently, spironolactone has been shown to be effective in managing resistant hypertension in the abdominally obese, so keep it in mind.
3. The white-coat effect and BP variability
Reassuring patients who appear to only have spikes of hypertension when they come to see you in the clinic may be the wrong thing to do. This recent Lancet article suggests that variability in BP, and spikes of hypertension in particular, may in fact be more dangerous than sustained hypertension. An approach to managing this problem is to avoid agents known to increase BP variability, such as beta blockers. Furthermore, patients who believe that it's okay for their BP to be elevated because they have recently been active need to be educated that this is incorrect.
4. Allopurinol for hypertension
Urate is periodically in and out of vogue as an instigator / perpetuator of kidney disease and hypertension. I personally believe it to be a marker of oxidative stress, but am still undecided about it having a direct role in kidney disease and progression. I have treated severe hyperuricemia in CKD patients with this specific intention / hope of attenuating disease. This recent JAMA paper suggests this approach is not complete madness. This short-term crossover study of adolescents with hypertension and hyperuricemia found that allopurinol 200mg bid controlled hypertension on it's own in 66% of patients. Another one to keep in mind
That's enough for today, I'll return to this topic next time.
Thursday, March 11, 2010
World Kidney Day 2010

Happy World Kidney Day! Celebrated every year in March, World Kidney Day is a day devoted to raising awareness of kidney disease across the globe. This year promised to be a huge success: cities throughout the world, from Bahrain to Belgium, held kidney awareness events, and celebrities such as Tom Hanks and French President Nicolas Sarkozy jumped on the kidney awareness bandwagon to lend their support to the cause. Besides raising awareness of our “amazing kidneys”, the day’s objectives were to encourage screening for CKD, recognize the importance of risk factors in the development of kidney disease, and encourage preventive behaviors. This year’s theme was “Protect Your Kidneys: Control Diabetes,” in order to place special emphasis on the growing prevalence of diabetic renal disease.
Tackling the problem of CKD is no small job. The last NHANES survey in 2009 estimates the U.S. prevalence of CKD stage II-V at 13%. While less prevalent in the developing world, CKD is expected to become a much greater problem due to the rapidly increasing rates of diabetes (an 85% increase in Central and South America, and 75% in the Western Pacific region, by 2025), not to mention hypertension. CKD has far-reaching effects on patient outcomes: for Medicare CKD patients in 2009, adjusted all-cause hospitalization rates were 1.5 times the non-CKD population, and all-cause mortality was twice as high as in non-CKD patients, even when adjusted for comorbid conditions. Then there is cost: in one article by DH Smith et al in 2004, the management of CKD alone was estimated to cost an additional $8000 annually per patient.
Wednesday, March 10, 2010
Lack of respect for the beans
Despite a list of absolute contraindications which include donors with active malignancy, untreated sepsis or active intravenous drug abuse, I have found that the limits are a little more subjective than expected, and with the organ shortage, transplant services are even more aggressive of using organs labeled as expanded criteria donor (ECD).
We had an interesting donor offer recently, in which a young female had died after a closed head trauma complicated by disseminated intravascular coagulation. The creatinine was normal and urine output was around 40cc/hour. Our transplant service decided to accept both kidneys. Not sure if you guys know, but the kidneys are actually the last organ to be removed from the deceased donor!? Liver, heart and lungs have preference!!! We had to wait almost 2 hours for another team to take out the liver before we were able to get to the beans… The concern was: could the DIC had damaged the kidneys?
DIC is associated with histopathological evidence of microthrombi in several organs, particularly in the brain, liver, lung, kidney and pancreas. A combination of mechanisms, including release of fat, phospholipids and thromboplastin from tissue—especially brain tissue—into the circulation, hemolysis and endothelial damage may promote systemic activation of coagulation. Cytokines also may have a role in the development of DIC.
The two recipients demonstrated a very slow increase in urine output postop associated with also small downtrending of the creatinine. A time zero biopsy showed multiple fibrin thrombi in the glomerular capillaries. As an important learning point, these allografts are usually associated with delayed graft function. Moreover, it is essential to ensure no significant necrosis is present on biopsy at time zero, before transplantation. It is believed that the recipient's own fibrinolytic system is able to slowly breakdown the thrombi and reestablish the renal function. Still, I was disappointed that the kidneys were last…
Tuesday, March 9, 2010
Hermansky-Pudlak Syndrome
 This is one that Nate would have liked....I recently saw a case of renal disease in an albino Peurto Rican woman with severe pulmonary fibrosis. She holds the diagnosis of Hermansky-Pudlak Syndrome (HPS).
This is one that Nate would have liked....I recently saw a case of renal disease in an albino Peurto Rican woman with severe pulmonary fibrosis. She holds the diagnosis of Hermansky-Pudlak Syndrome (HPS).HPS is an autosomal recessive disorder characterized by
- Oculocutaneous albinism
- A bleeding diasthesis (absense of dense bodies)
- Sometimes pulmonary fibrosis
- Sometimes granulomatous colitis.
The renal failure, pulmonary fibrosis, and granulomatous colitis are associated with lysosomal accumulation of amorphous lipid-protein complexes called ceroid lipofuscin. This is seen on the limited number who have undergone renal biopsy. The ceroid deposits accumulate in renal tubular epithelial cells of the proximal tubules with sparing of the glomerular epithelial cells and the distal tubular epithelium. Pathogenesis is still speculative and treatment options other than supportive care are currently unavailable.
Saturday, March 6, 2010
Compensation for living organ donation! A taboo no more but....

Talking to Nate always stimulated new ideas and new challenges. Last time I met him, we played soccer in the park with the kids (that was also a challenge but fun and easy to win…). We also talked about an interesting hot ethical topic (It was hard and no winners…). Compensation for living organ donors! A taboo no more but nonetheless remains an ethical dilemma that needs public debate, engaging mainly specialists who care for end-stage kidney disease patients. I thought using Nate’s blog for such a debate will please him, so here we are…
My approach will be, what are the facts?
- We have an increasing demand for kidney transplantation that our deceased donor waiting list can’t match. And living donation is not increasing especially when being an organ donor can negatively affect the likelihood of obtaining life, disability and health insurance. This concern is frequently raised by many potential donors.
- Recipients of living donor kidney transplants do better than recipients of deceased donor transplants.
- More people are dying on dialysis waiting for a transplant.
- Kidney donation is safe (Ibrahim et al. NEJM 2009)… Trust me, I finally watched a hockey game last week after I saw a Canadian flag in our Lab.
- And for the government (no offense) and the health insurance payers, Matas et al found that a LURD transplant saved $94,579 (US dollars, 2002), and 3.5 quality-adjusted life years (QALYs) were gained.
What are the ethical hurdles?
If we allow direct compensation between donors and recipients, we are turning the poor of our society into providers of body parts to wealthy people.
But what if the compensation is indirect through government agencies, providing donors, for example, with lifetime health insurance or tax deduction and maybe by treating them as the nation heroes which they deserve by the way.
I’ll be happy to hear your comments.
Friday, March 5, 2010
Familial renal glucosuria and a new drug for diabetes mellitus
 The kidney contributes to glucose homeostasis by reabsorbing ~180 grams of glucose daily. Two sodium-coupled glucose transporters play an important role in this process, SGLT1 and SGLT2. Of these two, SGLT2 (encoded by the gene SLC5A2 on chromosome 16p11.2) is the transporter which is predominantly expressed in the luminal brush border of the proximal tubule (S1 segment) and mediates primarily renal glucose re-absorption.
The kidney contributes to glucose homeostasis by reabsorbing ~180 grams of glucose daily. Two sodium-coupled glucose transporters play an important role in this process, SGLT1 and SGLT2. Of these two, SGLT2 (encoded by the gene SLC5A2 on chromosome 16p11.2) is the transporter which is predominantly expressed in the luminal brush border of the proximal tubule (S1 segment) and mediates primarily renal glucose re-absorption.Glucosuria, in the absence of diabetes mellitus, can occur in the setting of global dysfunction of the proximal tubule (PT), known as Renal Fanconi Syndrome. The latter is typically accompanied by excessive urinary excretion of amino acids, phosphate and bicarbonate. The occurrence of glucosuria in the absence of PT dysfunction and hyperglycemia is known as renal glucosuria or familial renal glucosuria (FRG) as it is recognized as an inherited disorder.
Patients with FRG have decreased renal tubular reabsorption of glucose from the urine in the absence of hyperglycemia or any other signs of tubular dysfunction. Glucosuria can range from 1 - 150 grams/1.73m2 per day. The majority of affected patients does not develop significant clinical problems and individuals are typically picked up during routine clinical tests. However, higher levels of glucosuria lead to osmotic diuresis and could cause volume contraction when access to fluid is limited.
Both, autosomal recessive and dominant pattern of inheritance for FRG have been reported. Mutations in SCL5A2 (encoding SGLT2) account for the majority if not all families with this condition. So far ~44 different mutations in 14 exons have been reported for FRG. Establishing a definite genotype-phenotype correlation has been difficult due to variable expressivity of SGLT2 and other genes that may have an overall impact on renal glucose reabsorption. In general, heterozygous mutations lead likely to mild glucosuria Here's a nice review.
In summary, FRG is a relatively benign condition nephrologists should be familiar with, especially with the availability of Dapagliflozin, a new drug for treatment of diabetes mellitus. First trials look promising since inhibiting SGLT2 appears effective and relatively safe.
Alimentary Azotemia?
 I am currently a clinical fellow, slogging through another consult month. As I am sure many of you have experienced, consult questions often come in batches. At least three times this week we have been called to see a patient with markedly elevated BUN out of proportion to creatinine with the team questioning uremia, need for renal replacement etc. In all three cases, there had been an associated drop in hematocrit, and so we suggested they consider an occult gastrointestinal bleed as a contributor to the azotemia. Having suggested the association several times this week, I decided to investigate the azotemia-GI bleed association further.
I am currently a clinical fellow, slogging through another consult month. As I am sure many of you have experienced, consult questions often come in batches. At least three times this week we have been called to see a patient with markedly elevated BUN out of proportion to creatinine with the team questioning uremia, need for renal replacement etc. In all three cases, there had been an associated drop in hematocrit, and so we suggested they consider an occult gastrointestinal bleed as a contributor to the azotemia. Having suggested the association several times this week, I decided to investigate the azotemia-GI bleed association further.For my first post....
Thursday, March 4, 2010
Creatinine: A Cautionary Tale
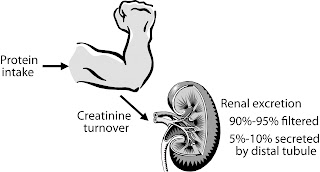 In recent months I've been seeing a number of referrals for patients with creatinine values in the normal range but eGFR as reported by the lab at below 60ml/min. This is particularly the case in middle aged women. Another group of cases troubling my Primary Care colleagues are male athletes with creatinine levels at or above the upper limit of normal but with no other apparent marker of kidney disease.
In recent months I've been seeing a number of referrals for patients with creatinine values in the normal range but eGFR as reported by the lab at below 60ml/min. This is particularly the case in middle aged women. Another group of cases troubling my Primary Care colleagues are male athletes with creatinine levels at or above the upper limit of normal but with no other apparent marker of kidney disease.Certainly this is reflective of the weakness of using creatinine and creatinine based estimates of GFR as our basis for determining renal function. Unfortunately there are limited clinically applicable alternatives. What do we tell these patients? Do they or do they not have kidney disease?
My approach is the following:
1. Authenticate renal function within the office practice to the extent possible.
2. Check Cockcroft Gault creatinine clearance
3. Check 24 hour or 12 hour urine creatinine clearance
4. Rule out rising creatinine within normal range by reviewing historical data if available
5. Rule out Microalbuminuria or Proteinuria as markers of intrinsic renal disease
6. Rule out CKD risk factors (Hypertension, Diabetes, etc)
7. Consider a renal U/S to evaluate renal size.
If all these data are within normal limits then I reassure the patient and suggest a 6 to 12 month follow up and will repeat the studies to confirm a normal picture. If there remains doubt an Iothalamate measured GFR needs to be obtained. The issue of estimated versus measured GFR is reviewed by Andrew Levey and Lesley Stevens in an excellent article in JASN last year.
For those athletes with elevated creatinine levels I recommend the same process making sure they: discontinue all NSAID use; do not take any creatine based dietary supplements; and limit their meat intake to reasonable levels before performing the testing.
David Steele MD.
Wednesday, March 3, 2010
Revealed: my "covert" project with Nate a.k.a TRP channels in the kidney
What are TRP channels? And why do they matter in terms of kidney health and disease?
TRP stands for Transient Receptor Potential, a large family of non-selective cationic channels with diverse tissue distribution and diverse cellular functions. They were initially discovered in Drosophila (fruit fly) photoreceptors where they mediate fly vision. Some refer to them as “the last bastion of ion channels” since there is still much we do not understand about their role in health and disease. There are a few TRP subfamilies, indicated by letters which are there for various historical reasons (a discussion for another day – just take my word for it for now, or refer to a review by Ramsey et al., Annu Rev Physiol. 2006;68:619-47), but there are the TRPCs, the TRPVs, the TRPMs, the TRPPs, the TRPMLs and the TRPAs.
A few fun facts:
- TRP channels (TRPV1) are responsible for the hot taste of chili peppers in your mouth.
- TRP channels (TRPM8) also give you the cold sensation of menthol.
But how are TRP channels pertinent to your kidneys? It turns out that they are actually involved in many aspects of kidney physiology and disease (called “channelopathies”):
We’ll start from my favorite: Proteinuric kidney disease. Mutations in TRPC6 were found in a number of families with adult onset Focal Segmental Glomerulosclerosis. Many of us are now actively involved in research to identify the precise mechanisms by which disease occurs in these patients, but deleterious effects due to channel mutations in glomerular podocytes are strongly suspected.
Another channel, TRPV5, mediates vitamin D dependent calcium uptake in tubular epithelial cells. The absence of TRPV5 channels (at least in mice) causes renal calcium wasting and bone loss.
Perhaps more intriguingly, mutations in TRPM6 have been implicated in human hypomagnesemia with secondary hypocalcemia (HGH) as this channel is involved in renal magnesium uptake.
And rather famously, of course, polycystic kidney disease (ADPKD) emerges due to mutations in two slightly more distant relatives in the TRP family, the polycystins, known as TRPP1 and TRPP2.
This of course brings us full circle back to Nate and his research, which was focused on cystic disease, and specifically cystic kidney disease.
As this is my first time blogging, please let me know what you think, your comments and suggestions are welcome!
New contributors to the Renal Fellow Network

I'd like to welcome the following new contributors to the Renal Fellow Network, who will begin writing this week. Between them, they have a wide range of clinical expertise and research interests. I'm confident that their contributions will really enrich and improve the network.
Dr. Albert Lam, Research Fellow Massachussets General Hospital
Dr. Anna Greka, Attending, Massachussets General Hospital
Dr. David Steele, Attending, Massachussets General Hospital
Dr. Ernest Mandel, Clinical Fellow Brigham and Womens Hospital
Dr.Hakan Toka, Research Fellow Brigham and Womens Hospital
Dr. Jamil Azzi, Research Fellow Brigham and Womens Hospital
Dr. John Niles, Attending, Massachussets General Hospital
Dr. Julie Paik, Research Fellow Brigham and Womens Hospital
Dr. Leo Riella, Research Fellow Brigham and Womens Hospital
Dr. Lisa Cohen, Research Fellow Brigham and Womens Hospital
Dr. Mario Rubin, Attending, Massachussets General Hospital
Dr. Mary Lieu, Research Fellow Brigham and Womens Hospital
Dr. Matthew Sparks, Research Fellow Duke
Dr. Melissa Yeung, Research Fellow Brigham and Womens Hospital
If any readers of the blog have topics they would like covered, please get in touch. Also, please keep the comments coming; they're always appreciated!
Saturday, February 27, 2010
A Man's Best Friend
 I'm sure many of you have a pet dog. Well, as Rover quietly licks himself in the corner, few would believe that he may hold the key to unlocking the genetic secrets of human disease, as suggested by this fascinating article in this weeks Nature Genetics.
I'm sure many of you have a pet dog. Well, as Rover quietly licks himself in the corner, few would believe that he may hold the key to unlocking the genetic secrets of human disease, as suggested by this fascinating article in this weeks Nature Genetics.Tuesday, February 23, 2010
Continuation of Renal Fellow Network
Monday, February 15, 2010
Nathan Hellman Rest In Peace
 It is with great sadness and sorrow that I am writing to inform you of the passing of Nathan Hellman, MD, PhD, and founder of the Renal Fellow Network Blog. I worked with Nathan in the Division of Nephrology of the Department of Medicine, Massachusetts General Hospital (MGH), Boston, where we were both fellows. He passed away on February 13, 2010 at the Massachusetts General Hospital after a short illness.
It is with great sadness and sorrow that I am writing to inform you of the passing of Nathan Hellman, MD, PhD, and founder of the Renal Fellow Network Blog. I worked with Nathan in the Division of Nephrology of the Department of Medicine, Massachusetts General Hospital (MGH), Boston, where we were both fellows. He passed away on February 13, 2010 at the Massachusetts General Hospital after a short illness. Nathan was most importantly a wonderful husband, father, son and brother. At work, he touched all of our lives with his warm heart and spirit, great sense of humor and remarkable intellect. He was an exceptional scientist, a talented and insightful clinician and a remarkably kind and humble human being. He is survived by his wife, Claire, his two children, Sophie and Max, his parents, Dr. and Mrs. Hellman, his two sisters, and their families.
A Memorial Service will be held to celebrate his achievements and commemorate his contributions to the MGH community at the O’Keefe Auditorium on Wednesday, February 17th at 2pm, followed by a reception in the Thier Conference Room at 3.30pm.
In lieu of flowers, memorials can be sent to:
Nathan Hellman Memorial Fund
PO Box 471044
Brookline Village, MA 02447
Wednesday, February 10, 2010
Tamm-Horsfall Protein and Kidney Disease

In this months issue of JASN, Kottgen et al. provide a nice proof of principle of the GWAS approach, by demonstrating urinary uromodulin levels predict incident CKD, in a prospective, case-control study of ten years duration. These findings were then externally replicated. In addition, they identify a protective SNP which confers a reduced risk of CKD via reduced urinary urimodulin concentration. Although the biology remains to be determined, this finding further supports the role of uromodulin in the pathogenesis of common variant CKD.
Tuesday, February 9, 2010
Did you know your endothelium is hairy?
 In the kidney, the glomerular endothelium is a little different than most other endothelia in that they contain fenestrae: actual pores in the endothelial cells which are large enough to let most proteins pass through, but small enough to exclude circulating blood cells. The glomerular fenestrated endothelium makes up a component of the glomerular filtration barrier (along with the glomerular basement membrane and the podocyte layers), and the classic teaching is that the negatively charged proteoglycans on this layer help form part of the charge barrier keeping albumin from getting filtered (though this is now being debated by some). Here are some pretty pictures of the glomerular fenestrated endothelium:
In the kidney, the glomerular endothelium is a little different than most other endothelia in that they contain fenestrae: actual pores in the endothelial cells which are large enough to let most proteins pass through, but small enough to exclude circulating blood cells. The glomerular fenestrated endothelium makes up a component of the glomerular filtration barrier (along with the glomerular basement membrane and the podocyte layers), and the classic teaching is that the negatively charged proteoglycans on this layer help form part of the charge barrier keeping albumin from getting filtered (though this is now being debated by some). Here are some pretty pictures of the glomerular fenestrated endothelium:
Monday, February 8, 2010
World Series of Poker Celebrity Kidney Transplant Recipient
 Meet Jennifer Harman. She is apparently one of the most famous female professional poker players in the world, having done quite well at the prestigious "World Series of Poker" tournament. It turns out she is also a kidney transplant recipient, having undergone 2 transplants. Admirably, she has become a real advocate for patients with kidney disease, seeking to raise awareness for living organ donation. Recently, she announced that she would be donating 1% of all profits to the NephCure foundation, which funds research relevant to nephrotic syndrome and FSGS in particular.
Meet Jennifer Harman. She is apparently one of the most famous female professional poker players in the world, having done quite well at the prestigious "World Series of Poker" tournament. It turns out she is also a kidney transplant recipient, having undergone 2 transplants. Admirably, she has become a real advocate for patients with kidney disease, seeking to raise awareness for living organ donation. Recently, she announced that she would be donating 1% of all profits to the NephCure foundation, which funds research relevant to nephrotic syndrome and FSGS in particular.
Sunday, February 7, 2010
Super Bowl Sunday Contrast Nephropathy Post
 How common is contrast nephropathy in the general outpatient setting? When you send one of your outpatients to get a CT scan with iv contrast, what is the real risk? Much of the randomized, controlled trial data on contrast nephropathy comes from INPATIENTS undergoing angiography or other procedures, often focusing on those with the greatest perceived risk of contrast nephropathy (e.g., diabetics, those with underlying CKD to begin with) in order to generate enough meaningful datapoints for analysis. An article by Mitchell et al in this month's CJASN reports the risk of contrast nephropathy in a mixed OUTPATIENT group--and finds that the risk of contrast nephropathy may actually be higher than what has generally been appreciated.
How common is contrast nephropathy in the general outpatient setting? When you send one of your outpatients to get a CT scan with iv contrast, what is the real risk? Much of the randomized, controlled trial data on contrast nephropathy comes from INPATIENTS undergoing angiography or other procedures, often focusing on those with the greatest perceived risk of contrast nephropathy (e.g., diabetics, those with underlying CKD to begin with) in order to generate enough meaningful datapoints for analysis. An article by Mitchell et al in this month's CJASN reports the risk of contrast nephropathy in a mixed OUTPATIENT group--and finds that the risk of contrast nephropathy may actually be higher than what has generally been appreciated.Saturday, February 6, 2010
Making Allograft Nephrectomies More Routine?
 As much as kidney transplant outcomes have improved over the decades, we're not perfect: a sizable group of patients end up transitioning to dialysis therapies after the kidney transplant stops working. What should be done with the allograft once you've given up hope for any meaningful allograft function? In many transplant centers, the prevailing wisdom is simply to leave the allograft in place, as long as it is not causing any exceptional systemic inflammatory response as a result of prolonged rejection. If nothing else, it avoids an additional surgery in a dialysis population that is often plagued with comorbidities and the potential for surgical complications.
As much as kidney transplant outcomes have improved over the decades, we're not perfect: a sizable group of patients end up transitioning to dialysis therapies after the kidney transplant stops working. What should be done with the allograft once you've given up hope for any meaningful allograft function? In many transplant centers, the prevailing wisdom is simply to leave the allograft in place, as long as it is not causing any exceptional systemic inflammatory response as a result of prolonged rejection. If nothing else, it avoids an additional surgery in a dialysis population that is often plagued with comorbidities and the potential for surgical complications.However, a recent study in this month's JASN by Ayus et al suggests otherwise, hinting that allograft nephrectomy might be considered as a more routine measure. The investigators probed the USRDS, looking at all transplant recipients who returned to long-term dialysis. A retrospective analysis demonstrated that those patients who had undergone an allograft nephrectomy had a 32% lower adjusted relative risk for all-cause death, even after controlling for factors such as age, comorbidities, and donor characteristics. The authors end the article by suggesting that "routine allograft nephrectomy in stable dialysis patients with a failed renal allograft should be evaluated against current management strategies in a randomized trial."
An accompanying editorial cautions against adopting this strategy too readily. They point out that patients in the allograft nephrectomy group tended to be younger and healthier, and even with appropriate adjustments to account for this, residual confounding and selection bias may well be altering the results. They also point to evidence that keeping the allograft in may actually be beneficial from an immune standpoint. Still, however, the retrospective by Ayus et al is intriguing and will doubtlessly be pursued further.
Friday, February 5, 2010
Warfarin and Vascular Calcification

Thursday, February 4, 2010
Drug Trials
 Check out the new poll question of the week in the right margin.
Check out the new poll question of the week in the right margin.
Wednesday, February 3, 2010
The trials of a lab rat...
 I was on the phone to one of my friends the other day. He's a renal fellow currently doing lab-based research in Boston. He's been having a difficult time of it lately, with none of his experiments working out, and has understandably been getting a bit frustrated and disillusioned.
I was on the phone to one of my friends the other day. He's a renal fellow currently doing lab-based research in Boston. He's been having a difficult time of it lately, with none of his experiments working out, and has understandably been getting a bit frustrated and disillusioned.Tuesday, February 2, 2010
The actin cytoskeleton of the podocyte
 Peter Mundel, a well-known researcher of podocyte biology, gave our Renal Grand Rounds today. Here's what I took away from this morning's talk:
Peter Mundel, a well-known researcher of podocyte biology, gave our Renal Grand Rounds today. Here's what I took away from this morning's talk:Monday, February 1, 2010
How Luminex Beads Work





















